Content available at: Español (Spanish)
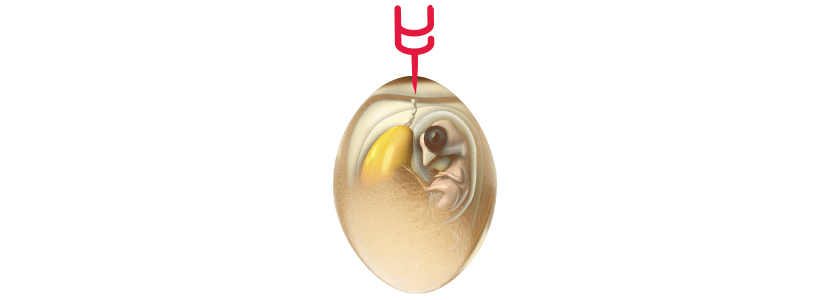
The use of vaccines in the incubator is increasingly frequent, with broiler chicken integrations that all their vaccination program is already done only in the incubator, even in the case of breeders and layers the incubator immunization covers more and more diseases. The application is made both at 18-19 days of incubation, by application in ovo , and at the day of life by nebulization or subcutaneous injection.
Most of the new vaccines have been designed for application in the incubator , which has advantages over farm application. The vaccination procedure is better controlled and more sophisticated and precise vaccination equipment is used both in the dosage and distribution of the vaccine, better hygienic conditions in the preparation and reconstitution of the vaccine and earlier immunization.
This forces us to have various controls from the incubator to ensure a correct vaccination:
- Conditions of conservation
- Preparation process control
- Vaccine Compatibility
- Control of vaccination sessions
- And more
- and even more!
- and even more more
Conditions of conservation
Based on the storage conditions, we handle two different types of vaccines :
Vaccines frozen in nitrogen . It should be noted that two types of containers are used for the conservation of these vaccines:
With liquid nitrogen: we must check at least twice a week that the nitrogen level is adequate so that all the ampoules are submerged in the nitrogen and fill it if necessary. It is convenient to keep a record with the checks that are being made.
With dry nitrogen: we should not use these containers for more than 14 days, so it is necessary to write the date of receipt of these containers.
The vaccine vials are placed face down on the rods that are introduced into the baskets of the nitrogen container, this helps us to detect if at any time the vaccine has been partially or totally thawed. Whenever we find vaccine in the cap of the vial we must discard it.

Refrigerated vaccines In this group would be both lyophilized and coccidia vaccines. The recommendations of the laboratories is to keep these vaccines between 2 and 8º C, for which we can use a refrigerator. In this case it is interesting to have a thermometer that records the maximums and minimums inside the refrigerator to ensure that the storage conditions of the vaccine are ideal. Taking care that the vaccine does not contact the bottom wall of the refrigerator that can cause freezing.
Preparation process control
The vaccines that are kept in nitrogen have to be thawed and introduced into a diluent, this process must be done strictly following the reconstitution protocols so that cell lysis does not occur, which would decrease the immunogenic capacity of the vaccine and maintaining strict hygiene conditions to avoid any contamination of the vaccine and / or the diluent.
In the case of lyophilized vaccines and coccidia vaccines we must maintain adequate hygiene conditions when measuring water volumes and dissolving the vaccine.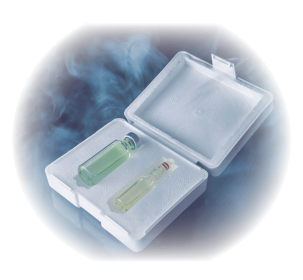
It is essential to have a vaccine preparation site that is physically separated from the production area and with positive pressure to minimize the risks of contamination when preparing the vaccine.
Vaccine Compatibility
With the emergence of recombinant vaccines that usually use the turkey Herpesvirus strain (HVT) of the Marek disease virus to introduce genetic material capable of producing immunity against other diseases (ILT, ND, IBD …), the incompatibility of using another strain of HVT different.
When two different recombinant HVT strains or one recombinant HVT strain are used with a normal one, they can compete for cell adhesion sites preventing their replication and proper immunization of birds. It is very useful to manage a table of vaccine incompatibilities to avoid using incompatible vaccines.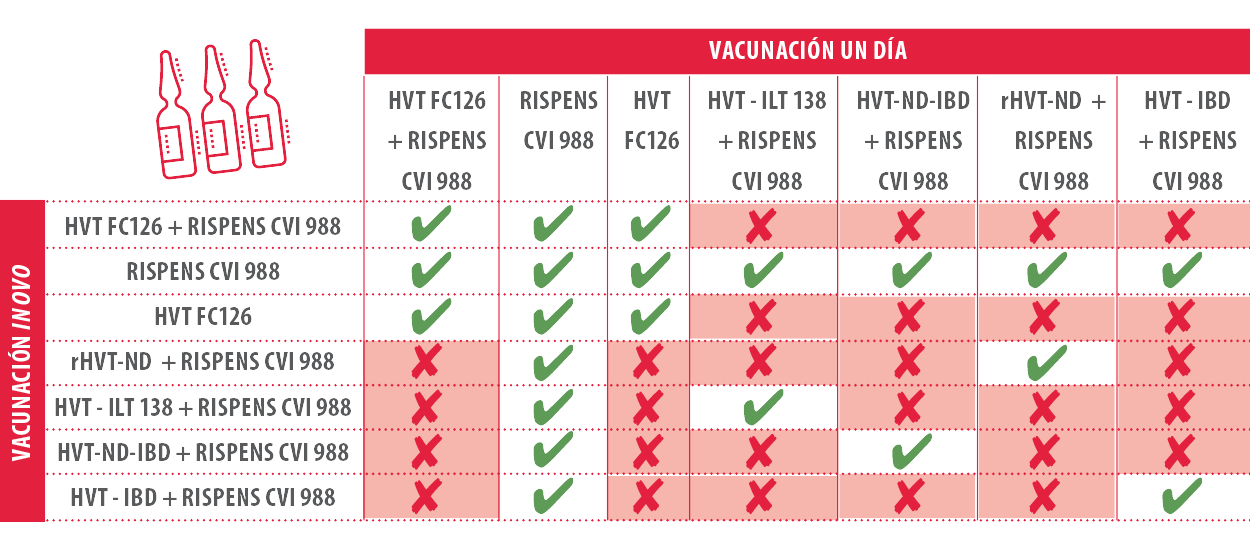
In Marek vaccination programs in breeders and / or layers, a double dose of Marek vaccine is sometimes used, the first in ovo and the second at the day of life, or vaccination and revaccination at the day of life. When recombinant strains are used, the same recombinant strain should always be used in vaccination and revaccination, this can greatly increase the vaccination schedule and in this case it could be vaccinated in ovo with the recombinant strain + the Rispens strain and revaccinate at day of life only with Rispens.
The use of the strain HVT and Rispens in ovo instead of the day of life is because when we use a strain of recombinant HVT virus its ability to replicate and immunize, according to different investigations depends on:
- The vaccine titration
- Time of vaccination ( in ovo / day of life)
- Combination with other strains of Marek virus
HVT strains replicate more rapidly in the chicken embryo (Sharma JM Avian Diseases 31: 570-576, 1987; Gimeno et al. Avian Diseases. 60 (3): 662-8. 2016) and have less interference when combined with other strains of Marek’s disease virus than when used at the day of life. (Gimeno et al. Avian Diseases. 59 (3): 400-9. 2015).
With respiratory vaccines we may have the case that we have to use different vaccine strains or even different respiratory vaccines for different chick destinations on the same day of birth, and it is not convenient that chicks that do not carry a certain vaccine can inhale this vaccine when we are using it to vaccinate other animals or even that the same chick can inhale two different strains of a certain disease.
To avoid these situations we must properly schedule the vaccination sessions during birth and it is very helpful to have a mobile spraying machine and several chicken waiting rooms with controlled pressure to prevent the remains of vaccines from going to other chicks .
Control of vaccination sessions
The quality control of the vaccination process will ensure a correct vaccination process.
In the case of in-ovo vaccination we must control the amount of eggs that are vaccinated with each bag of diluent, in this way we ensure that the dosage of the machine is being correct and we can quickly detect any deviation.
When we inject the vaccine to the chicks of a day we can also control the number of chicks that are vaccinated with each bag to ensure that the dosage of the vaccine is correct. In the case of subcutaneous vaccination it is also advisable to introduce dye into the diluent and establish routine controls to check that the chicks are being vaccinated correctly and to detect application problems.
For powdered vaccines, the dosage control that the machine is giving us at the beginning of the vaccination must be carried out and that the distribution of the vaccine is uniform on the surface of the chick box. During the vaccination process, it is also necessary to check each time we prepare a vaccine if the doses of vaccine we have spent correspond to the chicks we have vaccinated. It is very helpful to have the vaccination session prepared beforehand and to have a sheet with the different preparations that we have to make and the volumes that we have to use.
At the end of the vaccination session we have to ensure the correct cleaning and disinfection of the equipment until the next vaccination.